
Double Safety
with Safety Contact Sensor

with Safety Contact Sensor
1. Reliable Focused Ultrasound Mechanism for Face and Body treatment
2. Easy control and simple design with full function to treat face and body in one device
3. Possible to expand market from clinic to aesthetic shop
Doublo-m consists of 5 optimal cartridges according to different target areas wherever face and body. Various targeting depth of doublo-m cartridge satisfy different patients’ needs. Providing accurate treatment for both of face and body
| Cartridge | Target | Power | Spacing | |
|---|---|---|---|---|
| for Face |
 |
7Mhz /1.5mm |
Level 1-5 /Max. 0.65J |
Level 1-5 /Min. 1.2mm |
 |
7Mhz /3.0mm |
Level 1-5 /Max. 0.65J |
Level 1-5 /Min. 1.2mm |
|
 |
7Mhz /4.5mm |
Level 1-5 /Max. 1.4J |
Level 1-5 /Min. 1.5mm |
|
| for Body |
 |
7Mhz /8.0mm |
Level 1-5 /Max. 2.0J |
Level 1-5 /Min. 1.7mm |
 |
7Mhz /13.0mm |
Level 1-5 /Max. 2.0J |
Level 1-5 /Min. 1.7mm |

S.C.S (Safety Contact Sensor) whether a cartridge is perfectly contacted to skin which is naturally curved. Treatment shot is not working if the contact is not complete S.C.S ensure full skin contact and prevents any small scratch
Good grip and light handpiece relieve fatigue of practioners and slim-sized cartridge is suitable for arched face treatment
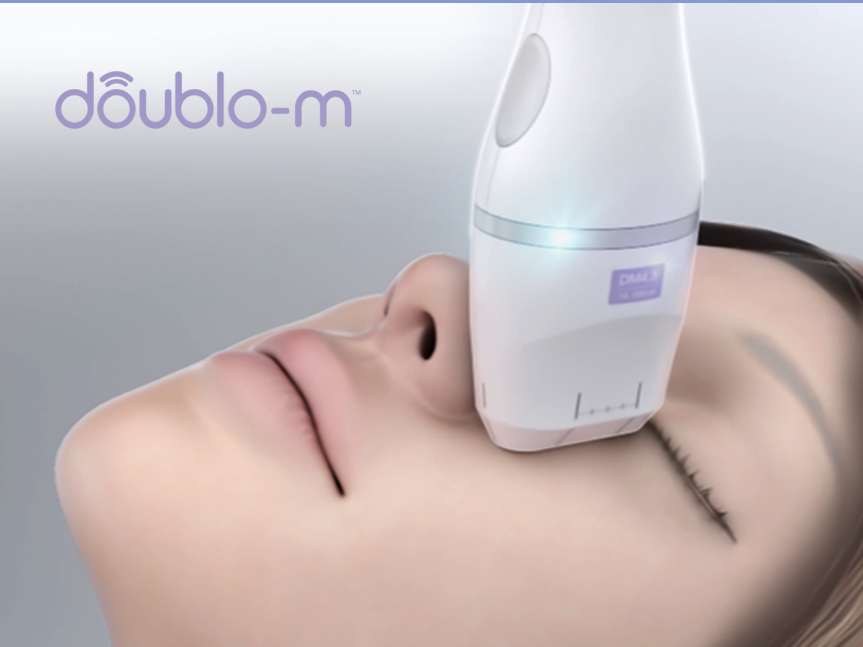
Two cooling chambers keep equal-beam quality for user to use stabilized energy, and our patented cooling system removes significant noise.

Blue light LCD lowers fatigue of the eyes. User friendly GUI is easy to understand even for the beginner. It also has alarming function to detect any malfunction
| Energy type | Focused Ultrasound |
|---|---|
| Fluence | 1~5 Level |
| Cartridge | FACE : DM1.5, DM3.0 DM 4.5
BODY : DM8.0, DM 13.0 |
| Spacing | 1.2 ~2.5mm (0.1mm/0.2mm Step |
| Length | Face cartridge 20mm, Body Catridge 25mm |
| Electrical Requirements |
AC 100-24OV, 50/60Hz |
| Dimensions | 341x420x254(WXDXH) |
| Weight | 5.5Kg (Main body) |

Dubai Derma 2019
Location : International Convention
Date : March 18 – March 20
HIRONIC participated at Dubai Derma 2019!
Regarding the exhibition, more than 200 visitors visited our booth and got many inquiries from 25 countries. Thanks to visitors, we could finish this exhibition very successfully, and it was a good opportunity to know the market trend and needs in the Middle East region and promote our new products such as Plasonic and Slimus. We sincerely hope to see you at Dubai Derma 2020 again.
Location : International Convention
Date : March 18 – March 20

HIRONIC participated at Dubai Derma 2019!
Regarding the exhibition, more than 200 visitors visited our booth and got many inquiries from 25 countries. Thanks to visitors, we could finish this exhibition very successfully, and it was a good opportunity to know the market trend and needs in the Middle East region and promote our new products such as Plasonic and Slimus. We sincerely hope to see you at Dubai Derma 2020 again.





KIMES 2019
Location : COEX, Seoul, Korea
Date : March 14 – March 17
HIRONIC participated at KIMES 2019!
HIRONIC successfully finished KIMES 2019 (35th Korea International Medical & Hospital quipment Show) held at the COEX Exhibition Center in Seoul from March 14th - 17th. KIMES is the largest medical equipment show in Korea and more than 70,000 visitors including 4,143 foreign visitors attended. Taking this opportunity, HIRONIC exhibited its main flagship products such as DOUBLO GOLD, MICOOL, SLIMUS, PLASONIC, ULTRA VERA, Q-FIT, and A-FIT. We were pleased to meet the medical professionals and share ideas with existing and potential partners. We are already looking forward to KIMES 2020 and welcome your visit.
Location : Seoul, Korea
Date : March 14 – March 17

HIRONIC participated at KIMES 2019!
HIRONIC successfully finished KIMES 2019 (35th Korea International Medical & Hospital quipment Show) held at the COEX Exhibition Center in Seoul from March 14th - 17th. KIMES is the largest medical equipment show in Korea and more than 70,000 visitors including 4,143 foreign visitors attended. Taking this opportunity, HIRONIC exhibited its main flagship products such as DOUBLO GOLD, MICOOL, SLIMUS, PLASONIC, ULTRA VERA, Q-FIT, and A-FIT. We were pleased to meet the medical professionals and share ideas with existing and potential partners. We are already looking forward to KIMES 2020 and welcome your visit.







ADD 2019
Location : WASHINGTON, D.C.
Date: March 1 - March 3
HIRONIC participated at AAD 2019!
HIRONIC attend AAD 2019 (American Academy of Dermatology) which is held at the Washington Convention Center from March 1st - 3rd, 2019. More than 18,000 doctors attended and, with this opportunity HIRONIC displayed its prime products such as SLIMUS, PLASONIC, Q-FIT, and A-FIT(USA FDA approved). We were glad to meet our current partners and also new potential partners and enjoyed the meetings with many medical professionals. HIRONIC is already looking forward to AAD 2020 to be held in Denver and welcomes your visit
Location : WASHINGTON, D.C.
Date: March 1 - March 3

HIRONIC participated at AAD 2019!
HIRONIC attend AAD 2019 (American Academy of Dermatology) which is held at the Washington Convention Center from March 1st - 3rd, 2019. More than 18,000 doctors attended and, with this opportunity HIRONIC displayed its prime products such as SLIMUS, PLASONIC, Q-FIT, and A-FIT(USA FDA approved). We were glad to meet our current partners and also new potential partners and enjoyed the meetings with many medical professionals. HIRONIC is already looking forward to AAD 2020 to be held in Denver and welcomes your visit








IMCAS 2019
Location : Paris, France
Date : January 31 – February 2
HIRONIC participated at IMCAS 2019!
IMCAS 2019 was another successful exhibition for HIRONIC! We had meaningful meetings with various medical professionals in field, and would like to thank everyone who visited our booth to share valuable time together. Not only that, we had a lecture about Doublo Gold and Plasonic, presented by Dr. Sabatier (Dermatologist, France) to provide clinical information about devices for users.
Location : Paris, France
Date : January 31 – February 2

HIRONIC participated at IMCAS 2019!
IMCAS 2019 was another successful exhibition for HIRONIC! We had meaningful meetings with various medical professionals in field, and would like to thank everyone who visited our booth to share valuable time together. Not only that, we had a lecture about Doublo Gold and Plasonic, presented by Dr. Sabatier (Dermatologist, France) to provide clinical information about devices for users.




EADV 2018
Location : Paris, France
Date : September 12 – September 16
HIRONIC will participate at EADV 2018!
EADV2018 will provide the best platform for all participants to boost and update their knowledge in dermatological science and technology through a variety of courses, workshops and lectures. Join HIRONIC to see innovative devices which includes our newly launched SLIMUS.
Location : Paris, France
Date : September 12 – September 16

HIRONIC will participate at EADV 2018!
EADV2018 will provide the best platform for all participants to boost and update their knowledge in dermatological science and technology through a variety of courses, workshops and lectures. Join HIRONIC to see innovative devices which includes our newly launched SLIMUS.



FACE 2018
Location : QEII Centre, London
Date : JUNE 14 – JUNE 16
HIRONIC participated at FACE 2018!
Hironic was able to successfully attend FACE 2018, UK’s premiere scientific conference featuring the latest clinical information, practical tips and updates in the field aesthetic treatment. It was our second launching show for SLIMUS and thankfully many doctors and professionals were interested in SLIMUS, and had done many demonstration treatments. For FACE 2018, HIRONIC displayed Doublo Gold, Micool-A, and SLIMUS. Once again, we highly appreciate your visit. See you soon!
Location : QEII Centre, London
Date : JUNE 14 – JUNE 16

HIRONIC participated at FACE 2018!
Hironic was able to successfully attend FACE 2018, UK’s premiere scientific conference featuring the latest clinical information, practical tips and updates in the field aesthetic treatment. It was our second launching show for SLIMUS and thankfully many doctors and professionals were interested in SLIMUS, and had done many demonstration treatments. For FACE 2018, HIRONIC displayed Doublo Gold, Micool-A, and SLIMUS. Once again, we highly appreciate your visit. See you soon!
하이로닉 영문 웹사이트로 이동합니다.
글로벌 웹사이트에는 수출용 제품으로
대한민국에서 허가하지 않은 장비 및 서비스
내용을 포함하고 있습니다. 해당 내용은
대한민국의 의료기기 광고 심의, 규제를 받지 않습니다.
It goes to the HIRONIC English website.
The Global Web Site contains equipment
and services that are not authorized by
the Republic of Korea These contents
are not subject to review and regulation
of medical devices in Korea.


하이로닉 EU 웹사이트로 이동합니다.
EU(Europe) 웹사이트의 내용은 "CE MDD 또는 CE MDR" 허가승인된 품목의 제품 정보에 관한 것으로, 대한민국의 소비자 보호를 위한 국내법 의료기기의 광고 심의, 규제를 받지 않습니다.
It goes to the HIRONIC EU website.
The contents of the EU(European) web page are about product information of items approved for "CEMDD or CEMDR" in EU countries. They are not subject to advertising deliberation and regulation of domestic law medical devices for consumer protection in Korea.


하이로닉 중국어 웹사이트로 이동합니다.
중국어 웹사이트에는 중국내 판매제품으로
대한민국에서 허가하지 않은 장비 및 서비스
내용을 포함하고 있습니다. 해당 내용은
대한민국의 의료기기 광고 심의, 규제를 받지 않습니다.
正在移动到HIRONIC的中文官网。
在中问官网内介绍的产品只在中国销售的产品,
不一定是在韩国得到认可和服务内容。
针对这内容没有受到韩国医疗仪器广告审议限制。
